Solubility in molten lithium carbonate of various oxides and calcium... | Download Scientific Diagram

Solubility of common oil field scales of injection water and high-barium concentration and high-salinity formation water | Semantic Scholar
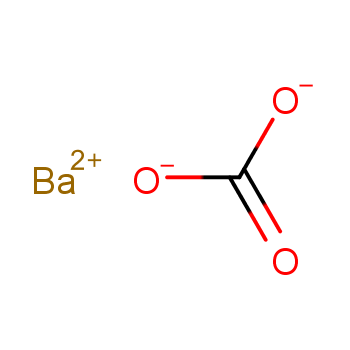
SOLVED: Calculate the solubility of barium carbonate, BaCO3, in pure water. Ksp = 2.0 × 10^-9 a. 2.2 × 10^-5 M b. 3.2 × 10^-5 M c. 1.3 × 10^-3 M d. 4.0 × 10^-18 M e. 4.5 × 10^-5 M

SOLVED: What is the molar solubility of barium carbonate in pure water if the Ksp of barium carbonate 5.1* 10-92 (lgnore any ion hydrolysis ) 2.6 * 10-17 1.7*10-3 5.1 x10-9 7.1*10-5 M None of these 11*10*3

Section 4: Solubility Equilibrium. Objectives Explain what is meant by solubility product constants, and calculate their values. Calculate solubilities. - ppt download