
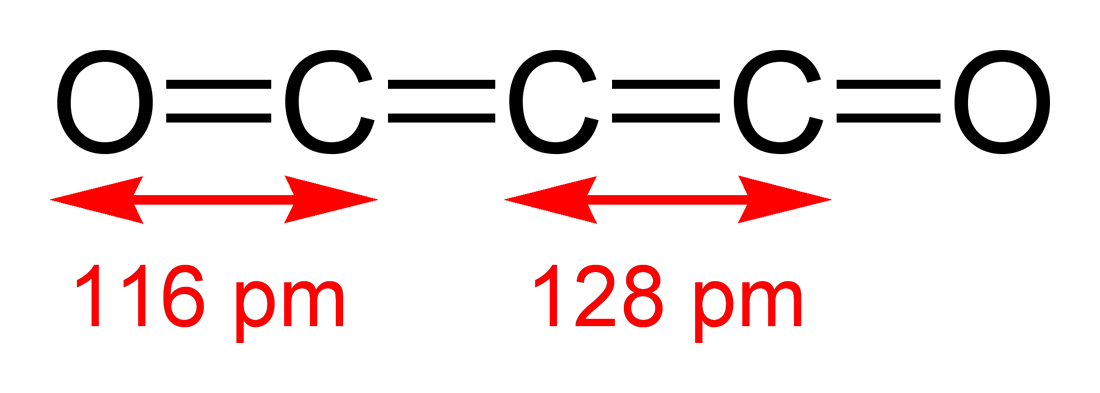
Carbon suboxide, C3O2, is an entirely linear molecule that is bonded in the order O-C-C-C-O. Draw the best possible Lewis structure for C3O2. Indicate any non-zero formal charges and resonance if appropriate.

Each carbon in carbon suboxide is :, (A) \( s p^{2} \)-hybridized, (B) \( s p^{3} \)-hybridized, (C...

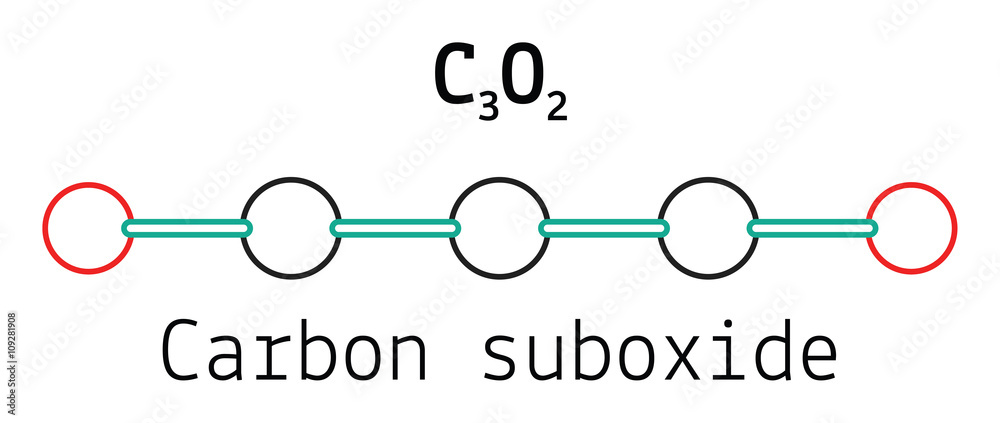
Carbon suboxide, or tricarbon dioxide, is an oxide of carbon. It is commonly described as an oily liquid or gas at room temperature with an extremely Stock Photo - Alamy











![Carbon Suboxide [C3O2] full Information || Oxidation State || Preparation || Properties || DC SIR Carbon Suboxide [C3O2] full Information || Oxidation State || Preparation || Properties || DC SIR](https://i.ytimg.com/vi/7bC1dedJhRA/maxresdefault.jpg)
