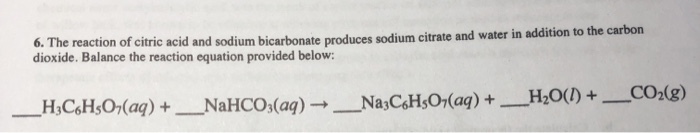Dsc studies on the decomposition of chemical blowing agents based on citric acid and sodium bicarbonate - ScienceDirect
Is the reaction bicarbonate with citric acid exothermic or endothermic? My teacher and my text book were saying it is exothermic, but I got results showing it is endothermic. - Quora

science chemistry compound citric acid sodium bicarbonate | Fundamental Photographs - The Art of Science
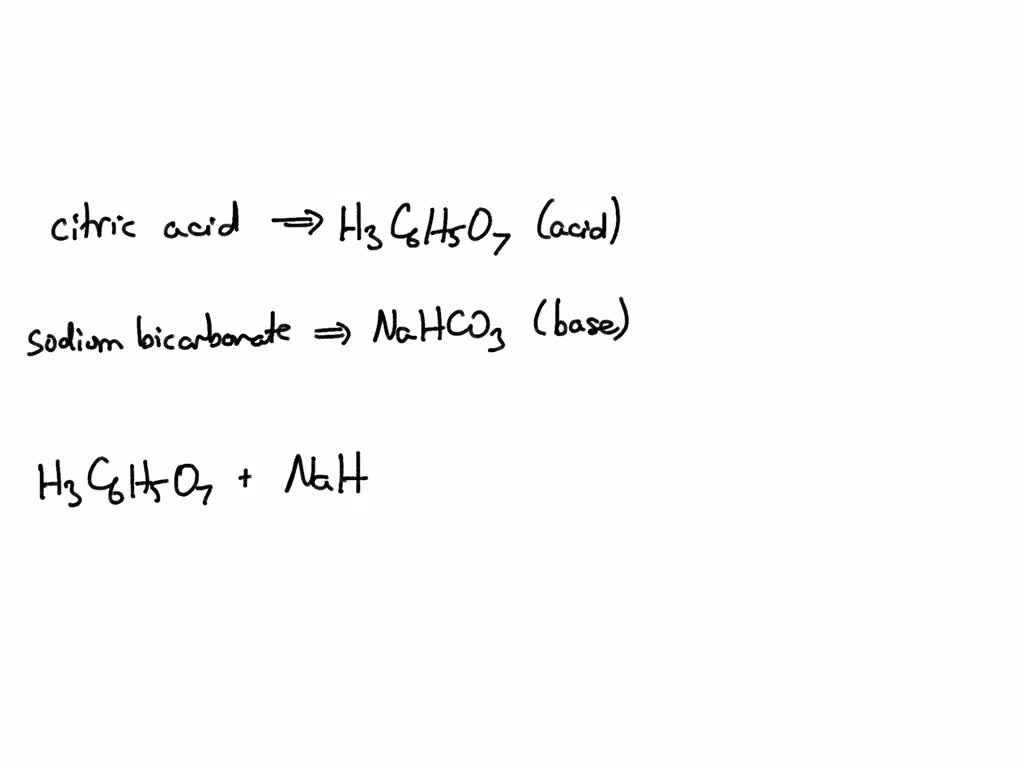
SOLVED: Write an equation for the neutralization of lemon juice (citric acid) with baking soda (sodium hydrogen carbonate or sodium bicarbonate). You equation should show a proton transfer to form carbonic acid

A pneumatic power source using a sodium bicarbonate and citric acid reaction with pressure booster for use in mobile devices | Semantic Scholar

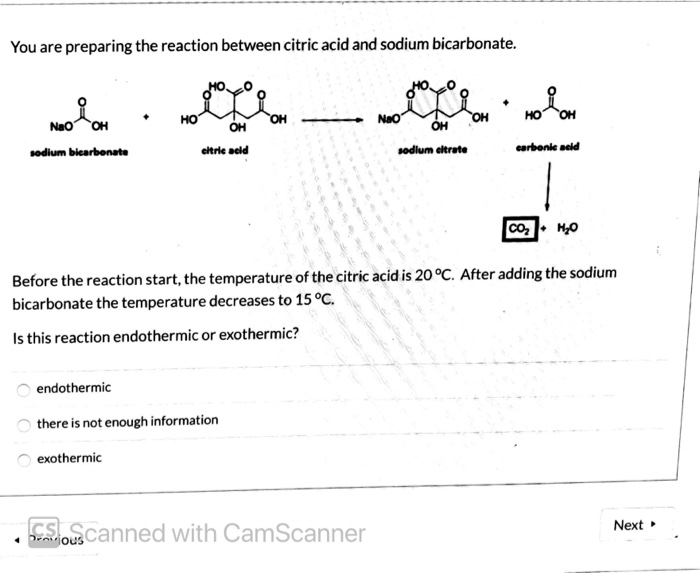
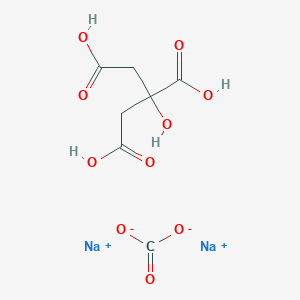
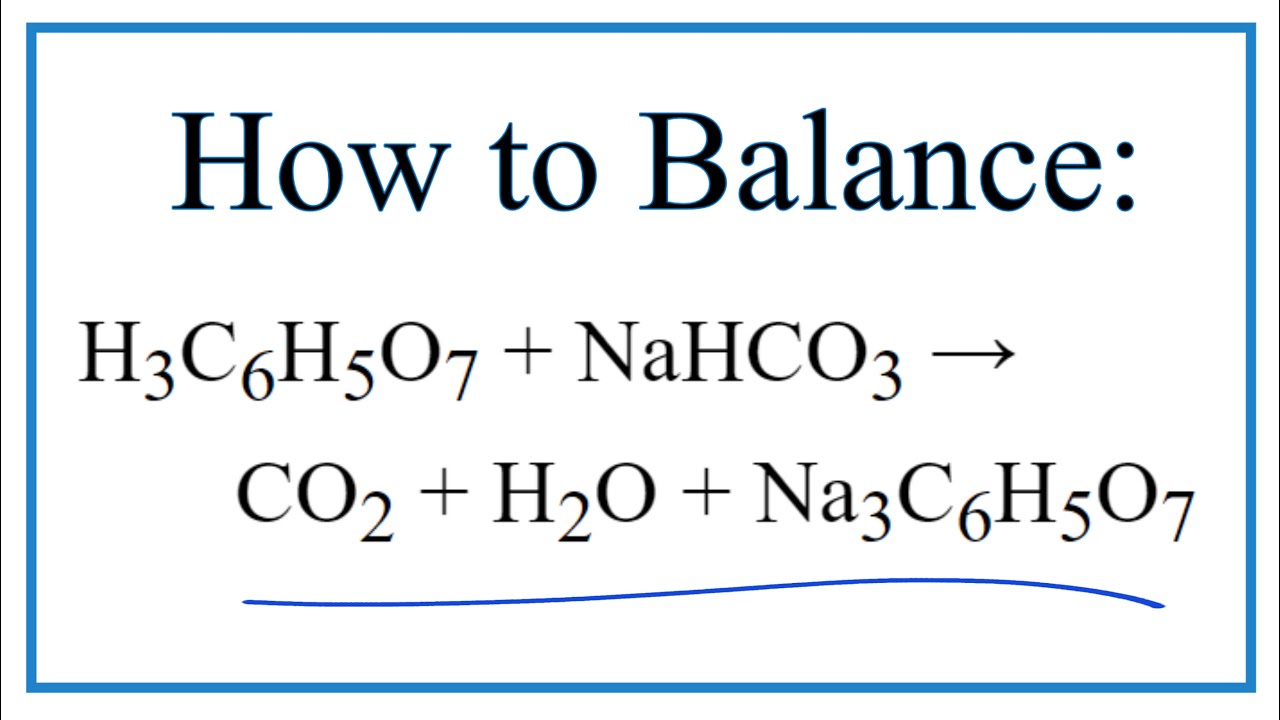
SOLVED: Aka-setter contains sodium bicarbonate (NaHCO3) and the citric acid (H3C6H5O7). When the tablet is dissolved in water, the following reaction produces sodium citrate (Na3C6H5O7), water (H2O), and carbon dioxide gas (CO2): (