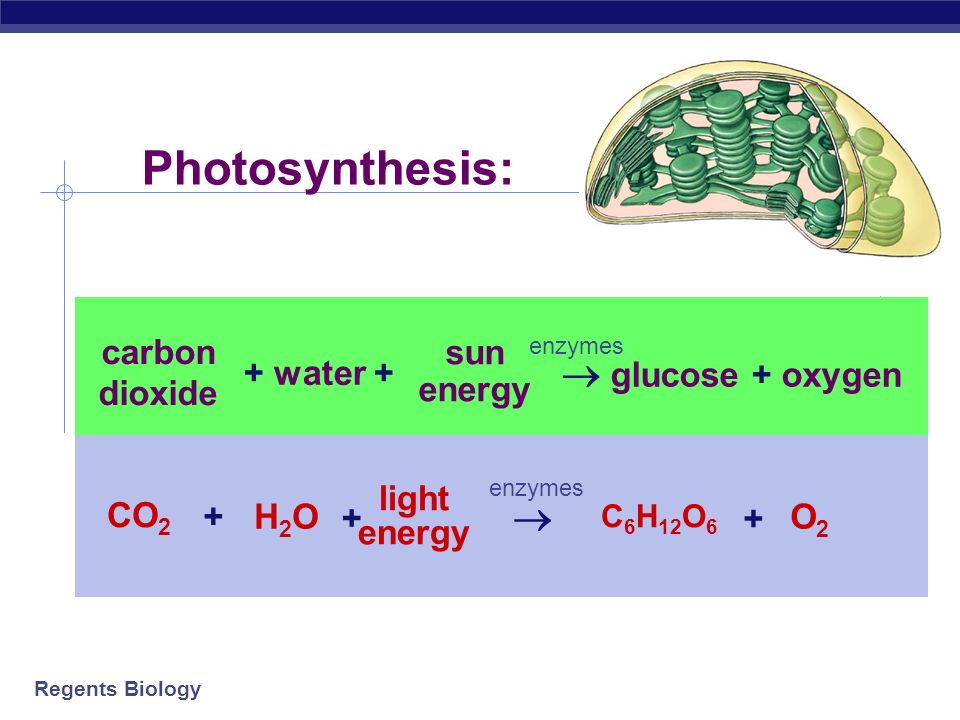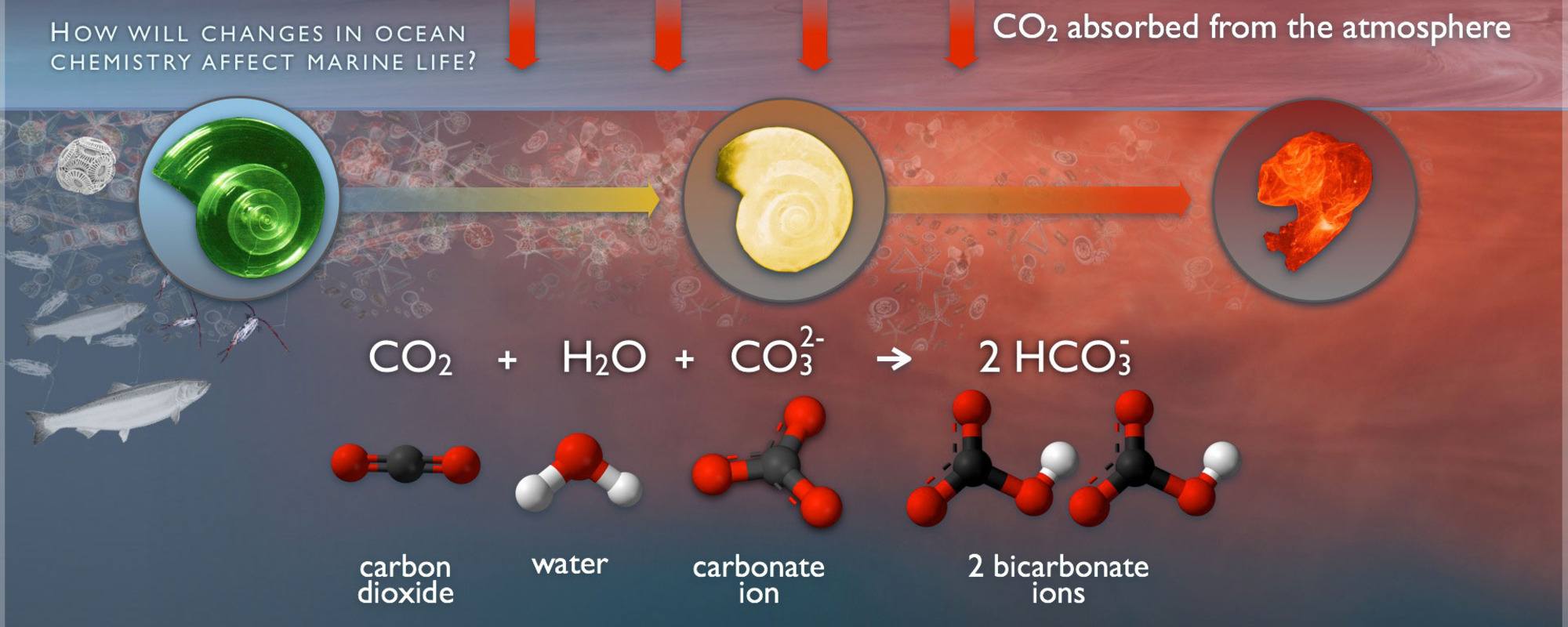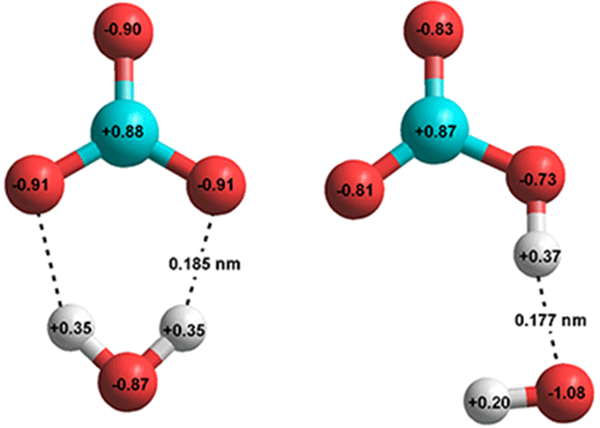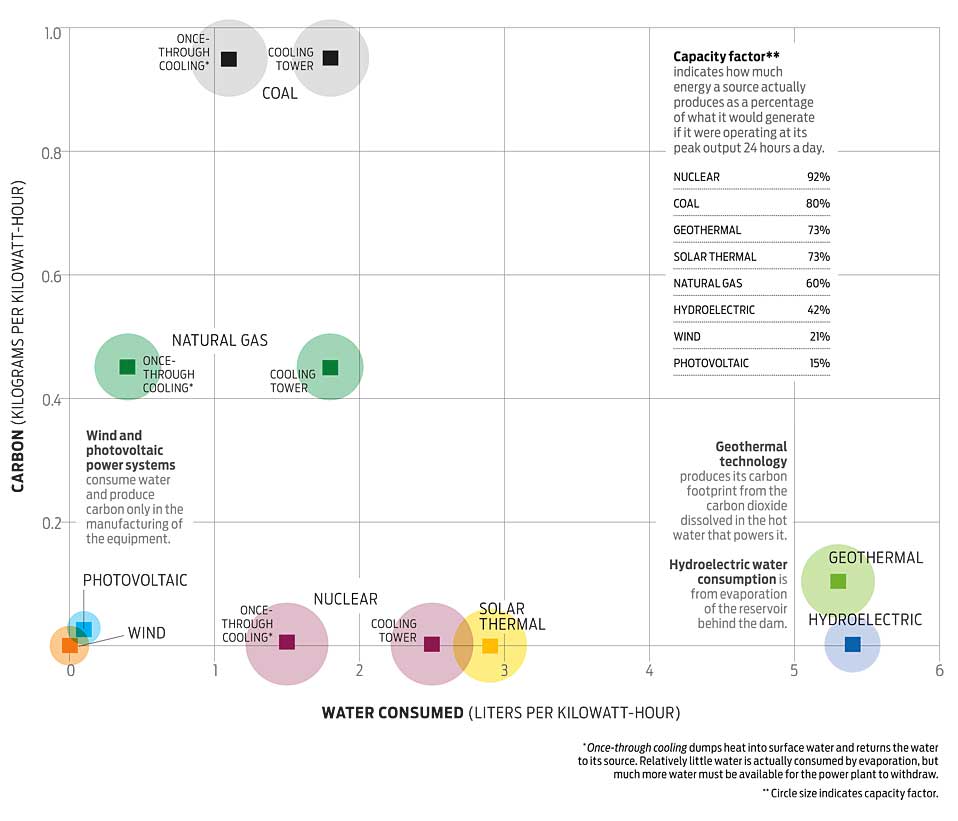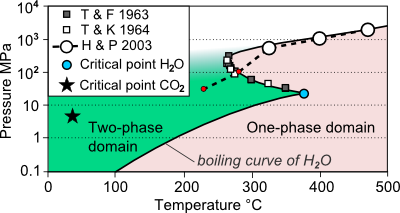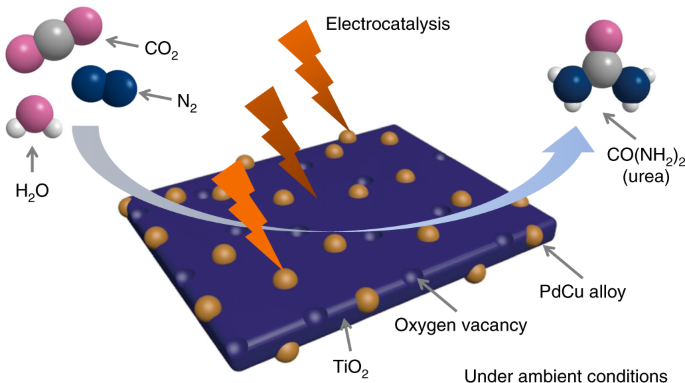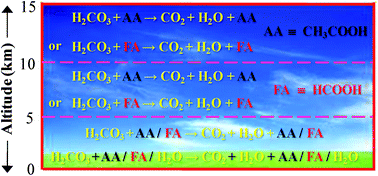
H2CO3 → CO2 + H2O decomposition in the presence of H2O, HCOOH, CH3COOH, H2SO4 and HO2 radical: instability of the gas-phase H2CO3 molecule in the troposphere and lower stratosphere - RSC Advances (

Reaction of CO, H2O, H2 and CO2 on the clean as well as O, OH and H precovered Fe(100) and Fe(111) surfaces - Catalysis Science & Technology (RSC Publishing)
![PDF] Effects of CO2 on H2O band profiles and band strengths in mixed H2O:CO2 ices | Semantic Scholar PDF] Effects of CO2 on H2O band profiles and band strengths in mixed H2O:CO2 ices | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/d35b84dc8965905b09b78fee990836ee8b77fcd6/6-Table5-1.png)
PDF] Effects of CO2 on H2O band profiles and band strengths in mixed H2O:CO2 ices | Semantic Scholar
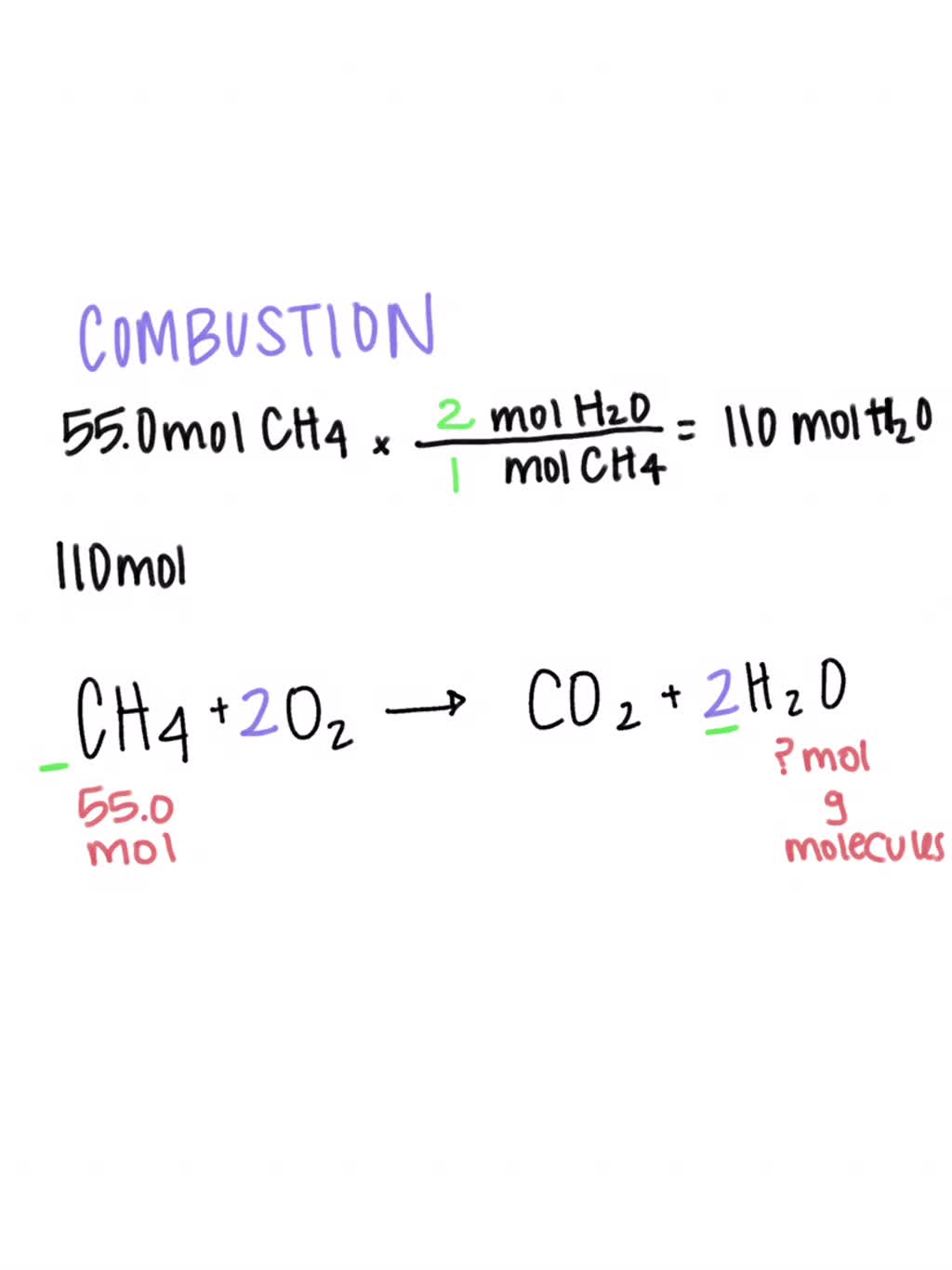
SOLVED: CH4 + O2 ——————- CO2 + H2O What type of reaction does this equation represent? Write and Balance the equation. Assume that there are 55.0moles of CH4. How many moles of
Write fully balanced equations for the following : (a) CO2 + H2O → ............ - Sarthaks eConnect | Largest Online Education Community

Water (H2O) and carbon dioxide (CO2) both have one central atom with two atoms bonded to it. However, one is a polar molecule and one is not. Draw the Lewis structure for
![Identify Bronsted - Lowry acids in the given reaction. [ Al (H2O)6 ]^3 + + H CO3^- [ Al (H2O)5 (OH^-) ]^2 + + H2CO3 A B C D Identify Bronsted - Lowry acids in the given reaction. [ Al (H2O)6 ]^3 + + H CO3^- [ Al (H2O)5 (OH^-) ]^2 + + H2CO3 A B C D](https://dwes9vv9u0550.cloudfront.net/images/4940114/ca6cb326-72a5-4e75-8a33-8b87ccbb46e4.jpg)
Identify Bronsted - Lowry acids in the given reaction. [ Al (H2O)6 ]^3 + + H CO3^- [ Al (H2O)5 (OH^-) ]^2 + + H2CO3 A B C D