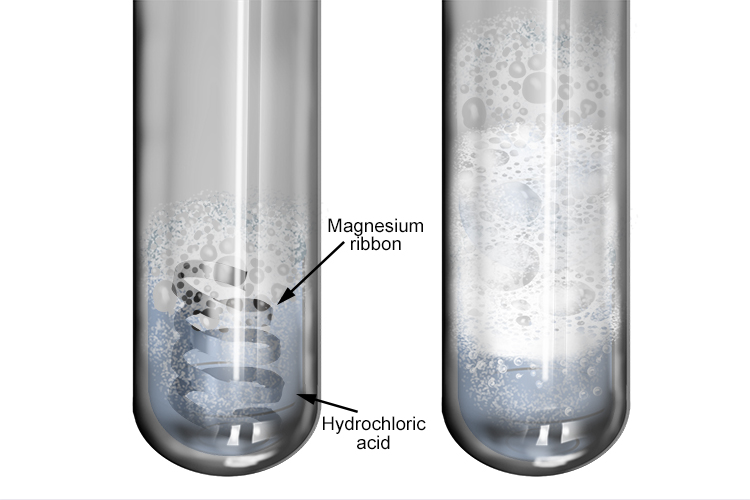
Magnesium reacting with acid. Magnesium (Mg) ribbon in a test-tube of hydrochloric acid (HCl). This reaction produces hydrogen gas (H2, fizzing) and a Stock Photo - Alamy
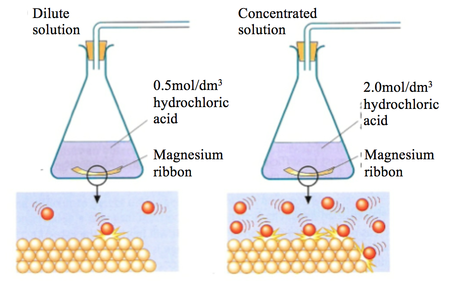
4.20 explain the effects of changes in concentration of solutions and pressure of gases on the rate of a reaction in terms of particle collision theory - iGCSE CHEMISTRY REVISION HELP
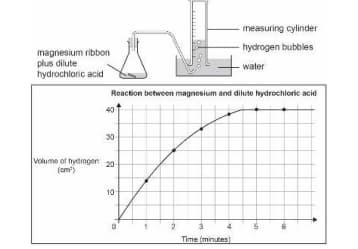
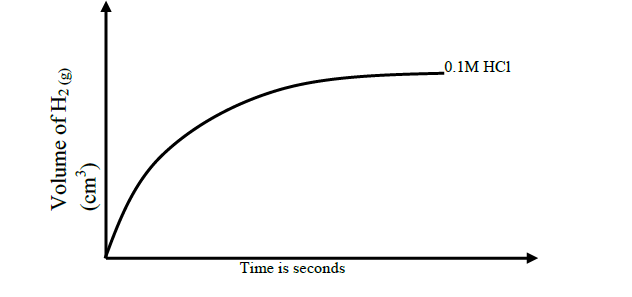
A piece of magnesium ribbon is added to a flask containing dilute hydrochloric acid Hydrogen gas is formed which is collected in the measuring cylinder The line on the graph indicates the

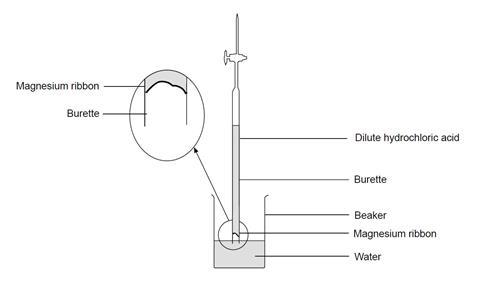
Describe an experiment to study the speed of reaction between dilute hydrochloric acid and magnesium, by measuring the volume of gas produced over time. - Study notes, tips, worksheets, exam papers

Bildagentur | mauritius images | Magnesium (Mg) ribbon in a beaker reacting with hydrochloric acid (HCl). This reaction creates magnesium chloride (MgCl2) and hydrogen gas (H2). Mg + 2HCl --> MgCl2 + H2